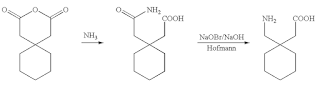
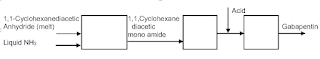
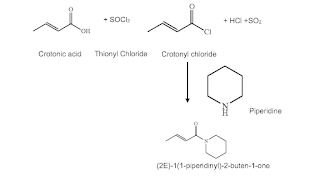
I am sure most of us have heard and/or read about “Green Chemistry (1998) ” (1), “Net Zero (2009)” (2) and then Decarbonizing” (3). Before we can tackle them we need to understand what do these mean and how they came about in chemical manufacturing processes needs to be understood. Before these words became in vogue environmental conservation has been the basic necessity of each process design and manufacture of every chemical but did not have high emphasis. Protecting our environment has been chemist and chemical engineer’s legacy but the degree has varied.
It is speculated that an investment of ~$9 trillion dollars (4) per year would be needed has to achieve 90% Net Zero or Decarbonization by 2050. What is the basis of this money, how it is going to be spent and how much of these monies are allocated to pharmaceuticals is an unknown. What methods and/or technologies will lead to decarbonization of API have not been proposed or even conjectured (5). The proposed yearly (~$9 trillion per year) amount is so large that It is beyond anyone’s imagination. I hope someone from McKinsey & Co. can shed some light on the numbers their breakdown and the technologies that will lead to decarbonizing of pharma APIs.
I am presenting my perspective and is not influenced by any “for profit or nonprofit” organization. It is also worth refreshing how did the industry arrive at the current situation. I have been involved in various capacities with environmental conservation in organic chemical (includes fine/specialty chemicals, additives, coatings and pharmaceutical and petrochemical industries since 1968. I believe there are simple ways to get to Net Zero in API manufacturing and their formulations but effort is needed. A concerted effort would need diligent application of principles of chemistry and chemical engineering. I am sure companies can achieve the target/s with lower spending. I am sharing methods that I have practiced and work. I have respectfully repeated myself to drive the point home. We have an excellent opportunity to reduce pharma’s carbon footprint.
HOW DID PHARMA GET TO THE CURRENT SITUATION?
Most of the chemical synthesis innovation started with the manufacture of dyes and colorants in Europe (6). Wood vats were used as they were available. These reaction vessels were open top or had wood lids. With time metallurgy developed. Processing equipment developed and mechanical agitators became the norm. As the mechanical engineering and metallurgy developed agitated vessels/reactors reaction vessels had lids and jackets that were necessary to heat and or cool the reactions other processing equipment (6, 7, 8) came about. With time process controls methodologies developed and have greatly assisted manufacturing processes.
Dye and colorant manufacturing companies (6) in Europe discovered many of the chemicals had disease curing value, they became the titans of pharma landscape (6). Companies also discovered that same organic chemistries applied to dye chemicals and disease curing chemicals. By changing the solvent amount used in the recipe their manufacturing processes could be fitted in the existing equipment. Cleaning of the equipment in between processes and products minimized cross contamination. This led to minimum new investment for the API (disease curing) synthesis chemicals. These practices have since continued.
While the chemicals were being developed, knowledge of chemistry and how manufacturing practices of the chemicals was being standardized. There were simultaneous developments in chemistry unit processes (9) and chemical engineering unit operations (10). Their adoption and application in the production of chemicals led to manufacturing process simplification and improvements in life style and life longevity. Products that improved life style became additives and the products that extended life were called pharmaceuticals. Interestingly their synthesis processes and equipment did not change except pharmaceuticals needed higher quality scrutiny as human lives depended on them.
Fitting processes in the existing equipment that started about 70+ years ago has led to excessive use of solvents for most of the API manufacturing processes and is a standard practice for API manufacture. “Art of fitting processes” has minimized capital investment for APIs and has become the pharmaceutical industry norm. This fits pharma’s business strategies for low yearly volume products that are needed for most of the APIs (one kilogram of API can produce ONE MILLION of one milligram tablets (10000 kilo converts to 100 million tablets). Practice of fitting processes has led to low ~about 35% equipment utilization (11). These practices have also led to highest organic emissions (solid, vapor and effluent) waste per kilo of product (12). Since the same equipment can be used to produce different APIs, government regulators came along and cGMP practices became mandatory. Cleaning in between each batch and different products is necessary to prevent cross contamination of products. Excessive amounts of solvents are needed to fit the process in the equipment for the highest conversion they can achieve has brought us to the current situation where global warming is knocking on our doors. As explained later creating an environmentally friendly process is the NEW challenge for the pharmaceuticals. We have the wherewithal that is needed but have to master it. I have been fortunate as I practiced environmental conservation for the last 55+ years and have been mindful.
PATHWAYS TO EMISSION REDUCTION IN FINE/SPECIALTY/ADDITIVES/COLORANTS (LIFE STYLE) AND PHARMACEUTICALS: API (DISEASE CURING CHEMICALS) AND THEIR FORMULATIONS:
Chemical industry and that includes life extending (API) and life style improving (additives) products do not need to name existing methods as new manufacturing technologies (13) but apply the fundamentals of science and engineering that are taught to us differently. Laboratory practices teach us the reaction chemistry (9). How we translate them to produce a product (10) is up to each chemist and chemical engineer. As suggested earlier environmental conservation has been part of chemical engineer and chemist’s teaching legacy but their degrees of application has varied. Yearly production volume of each chemical dictates the manufacturing process for each chemical i.e. would they be a batch (14) or a continuous (15) process. Their definitions have been established and are over 100 years old. Calling a batch process a continuous process has become in vogue lately. Continuous processes are product specific.
OUR LEARNINGS AND TEACHINGS: FITTING THE PROCESS IN THE EXISTING EQUIPMENT:
Since the inception of the fine/specialty/coatings/additives and pharmaceuticals industry (active pharmaceutical ingredients (API) and their formulations) chemists and chemical engineers as stated earlier have mastered the science and engineering of fitting their processes in the existing equipment. This tradition started in earnest about 70+ years ago when chemical companies dyes, colorants and other chemicals found that some of their molecules could cure diseases (6,7,8). This is just a reminder to us all.
PATH TO GET TO NET ZERO:
In the fine/specialty chemicals and their younger cousin API and their formulations show us pathways (9,10) that we are taught but have not exploited totally. We start with the synthesis in round bottom flask. In my own way I call commercial reactors larger round bottom flasks that have built in heating and cooling systems. This is due to the process developers taking a short cut of fitting chemistries in the existing equipment where various unit operations can executed. Reason for this path has been our laboratory teachings and availability of existing plant equipment. Unless we are proactive (16) to get to Net Zero, it is very likely that the chemical industry and which includes API and fine/specialty chemicals, will not make much progress. Out of the box thinking is needed. It has to be a collaborative effort and village’s (6,7) help is necessary.
Each chemical optimally reacts with the selected chemical to produce the desired product. Physical chemistry (17), unit processes (9), reaction kinetics (18) and unit operations (10) show/teach us how mutual behavior of chemicals can be capitalized on to create excellent and simple processes (6,7,8, 19, 20, 21, 22, 23, 24, 25, 26, 27). If the pharmaceuticals (API) need to get to Net Zero they will have to collectively capitalize on physical properties (16) of chemicals used and produced, unit processes (9) and unit operations (10) differently. Such processes compared to their current processes would be generally get lot closer to NET ZERO. If the pharmaceutical industry cannot get to NET ZERO outliers like Steve Jobs (28, 29) would be needed at every company. In addition giving new names to existing methods is not going to change the landscape (30). Industry will have to be proactive.
I am emphasizing it again that understanding and exploitation of physical and chemical properties along with reaction kinetics (18), thermodynamics (31), unit processes (9) and unit operations (10) give us opportunities to scale up and commercialize many chemical processes using smaller sized equipment. That immediately leads to solvent reduction per kilogram of the product. Net Zero happens naturally. These are exhilarating experiences.
CAPITALIZING ON MUTUAL BEHAVIOR OF CHEMICALS:
Why we have not capitalized on mutual behavior is an intriguing question and the answer may be simple. We are taught fundamentals of chemistry and chemical engineering but may not be exposed “how to manipulate and capitalize on them to create excellent processes”. That comes only with hands on experience or when we see examples of how one capitalizes on mutual behavior to create simple and excellent process. They generally are eureka moments. In many cases this results in solvent reduction and/or reaction simplification through reduced time and process simplification.
Knowledge (physical properties of reactants, their mutual behavior, unit processes, unit operations and understanding of various available processing equipment) would be applied to according to production volumes for the existing and new products. Creativity and imagination can simplify many processes at minimum cost and/or investment. I have emphasized many times over to drive a point that we have opportunities to get to “Net Zero”.
Learning curve to capitalize on mutual behavior of chemicals used and produced in every reaction is generally short as every chemist and chemical engineer knows the fundamentals of chemistry and chemical engineering of each product they produce. They have to be creative and imaginative to apply them. However, many would likely say “it can’t be done”. Saying “NO” is the simplest answer to anything different from what we are used to . This is human nature. Examples of how mutual behavior can be capitalized and are shared (6, 7, 8, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43, 44, 45, 46, 47,48, 49).
Yes, again a totally different thinking and execution of manufacturing processes will be necessary. Basically to minimize solvent use per kilo of each product requires capitalizing on the mutual behavior of solution or slurry of each raw material, intermediate and finished product. Process is very simple. As soon as the chemistry of each product is defined it is necessary that the village (6, 7, 8) gets involved to develop, design and commercialize the product. This is a deviation from current practices. Knowledge of physical and chemical properties and their mutual behavior specially solubilities have to be well known. If they are not known they will have to be developed. The whole process development has to be internalized. This is extremely helpful during the life of the product for process design, changes and any troubleshooting of the manufacturing process and changes. Underlying question we have to ask ourselves is “what can be done to have an all liquid process”. This will minimize using inert liquids as solvents, a major step towards “Net Zero”.
A simple example would be a low melting solid starting material could be dissolved in a solvent, solution made and fed to a reactor at e.g. 40% concentration vs. fed as liquid at 100% concentration in an inline circulating loop to finish the reaction. There are many examples of such reaction systems in the references (6,7,8). Not only the solvent can be was minimized but possibly a batch process could be converted to a batch campaign process or a totally continuous process. Experienced chemical engineers can further simplify processes by using modular and/or equipment (6,7,8) that may exist on their sites. Pilot plant and its equipment can be a gold mine. If done right some of the products could be that some of the APIs that have consolidated global volume of about 100,000 kilos per year or more could consolidate and be produced using continuous process rather than batch processes at multiple sites.
ISSUES THAT WOULD HAVE TO BE DEALT WITH:
One underlying question for the pharmaceutical industry is that it is very aware of having the highest emissions per kilo (12) of product in the chemicals related industries but it has not been proactive to improve its manufacturing technologies to lower them. Is it the costs involved in lowering emission through commercializing better processes and not getting the desired return or the regulatory hurdles that are expensive that prevent companies to make an attempt? I don’t want to speculate the answer and let the pundits cogitate over the issue. Whatever the answer, pharmaceutical industry has to deal with the issue that has been overlooked. It cannot procrastinate any longer.
There are challenges. We can call them “ROADBLOCKS” to get to Net Zero. They are different for brand and generic products. Even with some commonalities most likely each will have different pathways. All of the necessary work to get to NET ZERO has to be done at internally at each company as the chemists and chemical engineers are most familiar with their products and processes. They are the product and process experts (6, 7, 8). Outsiders may be able to guide them but the detailed work has to be internalized. Outsourcing work, it is my guess, will extend time as the learning time will further delay the process. I don’t think companies want to share their family jewels. It is best to discuss the brand and the generic products separately.
NEW BRAND AND NEW GENERIC PRODUCTS:
I am not going to go in the details but everyone who is involved in Net Zero should understand that the process starts when the chemistry is written on paper i.e. the process starts with inception of the product. Village (6,7,8) has to get involved right away as if the product is going to be a success and commercialized. Even if it is not, it is an excellent opportunity to known the chemicals used and produced. It will be a learning opportunity for how the process equipment can be manipulated to simplify processes. Engineers can think, scout and tinker with the different processing equipment and cross fertilize from different industries. Every effort has a common goal and that is minimize solvent use and produce the highest quality product. This is a significant deviation from current practices. As indicated earlier even if the product is not commercialized, it will be a team effort and useful for future projects. This will be total landscape change with significant resistance.
EXITING BRAND AND GENERIC PRODUCTS:
“Net Zero” pathways for the existing brand and generic products will be different in many ways but will also be similar. Who is going to invest and what would be the return and its value will be a major concern and question. If there is no product cost reduction, which means no payout, no one will pay for the investment. If company’s costs go up even after solvent reduction and they get passed on to the patients i.e. distributors keep their profits, overall drug prices will go up and there will be mass uproar against “Net Zero”. Every such program could come to screeching halt. Way out of this dilemma could be direct sales to patients and eliminating the current distributors and let the competition dictate the sale price. Competition will define the new landscape. Current pharmacy benefit managers (PBM) will not like that and will interfere for “Net Zero” becoming a reality. In US Congress would have to get involved. Likelihood of that happening is minimal to none.
Regulators could demand bioequivalence of products from alternate processes. Proving that could be expensive and time consuming. Brand companies would not do anything for the products currently under patent unless they assure product performance with new processes. Regulators will have the final say.
It is very likely that in its effort to achieve API manufacturing landscape could change. This could happen due to the needed investment, regulatory expenditures, pressures, pharmacy benefit managers (PBMs) and realignment of the manufacturing and changing landscape. There may be additional questions. If 50% to 60% reduction can be achieved in the next 15 years, it will be a major accomplishment. I have made an attempt that I believe would lead to “Net Zero”. It is a start and will need fine tuning. Doing something is better than doing nothing. Please share your knowledge. It is an opportunity to preserve our planet “EARTH”. Thank you.
Girish Malhotra, PE
EPCOT International
References:
1. Green Chemistry: https://www.acs.org/greenchemistry/what-is-green-chemistry.html Accessed September 15, 2024
2. Net Zero: https://eciu.net/analysis/infographics/net-zero-history
3. Decarbonizing: https://www.unepfi.org/grt/2016/programme/decarbonizing-finance/ Accessed September 10, 2024
4. What is Net Zero: McKinsey & Co. Accessed November 2022
5. Witte C. et. al. Decarbonizing API manufacturing: Unpacking the cost and regulatory requirements, July 26, 2024 McKinsey & Co. July 31, 2024
6. Malhotra, Girish: Active Pharmaceutical Ingredient Manufacturing: Nondestructive Creation De Gruyter April 2022
7. Malhotra, Girish: Chemical Process Simplification: Improving Productivity and Sustainability John Wiley & Sons, February 2011
8. Malhotra, Girish: Chapter 4 “Simplified Process Development and Commercialization” in “ Quality by Design-Putting Theory into Practice” co-published by Parenteral Drug Association and DHI Publishing© February 2011
9. Shreve, R. N. Unit Processes in Chemical Engineering, Industrial and Engineering Chemistry,1954, 46, 4, pg., 672, Accessed June 22, 2020.
10. McCabe W. L & Smith J. M. Unit Operations of Chemical Engineering McGraw-Hill Book Company Second Edition 1967
11. Schrader, Ulf: McKinsey & Co. Operations can launch blockbuster in pharma, February 16, 2021
12. Sheldon R.A. The E factor 25 years on: the rise of green chemistry and sustainability, Green Chemistry https://pubs.rsc.org/en/content/articlelanding/2017/gc/c6gc02157c/unauth#!divAbstract , 2017, 19, 18-43 Accessed February 17, 2021
13. Malhotra, Girish: Manufacturing Technology Innovations in Pharmaceutical Manufacturing, Profitability through Simplicity, February 23, 2024
14. Batch Process, https://en.wikipedia.org/wiki/Batch_production Accessed June 1, 2019
15. Continuous Process https://en.wikipedia.org/wiki/Continuous_production, Accessed June 1, 2019
16. Malhotra, Girish: Why Fitting a Square Plug in a Round hole is Profitable for Pharma and Most Likely Will Stay? Profitability through Simplicity August 1, 2014
17. Levine, Ira: Physical Properties Sixth Edition 2009 McGraw Hill
18. Levenspiel, O. Chemical Reaction Engineering, John Wiley & Sons Inc, 1972, Second Edition, Accessed December 20, 2020
19. Sociochemicology Sociochemicology 2013
20. Malhotra, Girish: Secret Life of APIs , Profitability through Simplicity, Feb. 13, 2013
21. Malhotra, Girish: Rx for Pharma, Chemical Engineering Progress, 105 (3) March, 34-38 (2009),
22. Malhotra, Girish: Focus on Physical Properties To Improve Processes: Chemical Engineering, 119 (4), April, 63-66 (2012).
23. Malhotra, Girish: Conflicts and opportunities in the Life of APIs, Chimica Oggi-Chemistry Today Vol. 31 (4) July/August 2013 pgs. 10-13
24. Malhotra, Girish: Focus on Physical Properties To Improve Processes: Chemical Engineering, Vol. 119 No. 4 April 2012, pgs. 63-66,
25. Malhotra, Girish: Process Simplification and The Art of Exploiting Physical Properties
Profitability through Simplicity March 10, 2017
26. Malhotra, Girish: Capitalizing on Mutual Behavior and Chemical Reactivity of Chemicals, Profitability through SimplicityMay 29, 2023
27. Malhotra, Girish: Chemicals tell us how to exploit their behavior for better processes. Clues are ignored. Should we? Profitability through Simplicity, June 20, 2023
28. Malhotra, Girish: Can the Combination of Creative Destruction and “Steve Jobs’ Traits” Lead to Pharma QbD Spring? Profitability through Simplicity, April 15, 2012
29. Malhotra, Girish: Does the Pharmaceutical Industry Need A Steve Jobs? Profitability through Simplicity, November 8, 2011
30. Malhotra, Girish: Is the New Terminology Going to Make the Pharmaceutical Processes Environmentally Friendly and Economic? Profitability through Simplicity, June 5, 2012
31. Dodge, B. F. Chemical Engineering Thermodynamics, McGraw-Hill Book Company, 1944
32. Malhotra, Girish: Quick Review of Chemicals Related Process Development, Design and Scale up Considerations, Profitability through Simplicity, November 7, 2018
33. Malhotra, Girish: Considerations to Simplify Organic Molecule (API) Manufacturing Processes: My perspective, Profitability through Simplicity, April 20, 2019
34. Malhotra, Girish: Active Pharmaceutical Ingredient Manufacturing (API) and Formulation Drive to NET ZERO (Carbon Neutral)? Profitability through Simplicity, April 29, 2021
35. Malhotra, Girish: Climate Change and Greening of Pharmaceutical Manufacturing, Profitability through Simplicity, January 24, 2022
36. Malhotra, Girish: Pharma’s Active Pharmaceutical Ingredient Manufacturing: Their Environmental Impact and Opportunities, Profitability through Simplicity, March 3, 2022
37. Malhotra, Girish: Art and Science of Chemical Process Development & Manufacturing Simplification https://www.aiche.org/ili/academy/webinars/art-and-science-chemical-process-development-manufacturing-simplification May 17, 2023
38. Malhotra, Girish: Chemicals tell us how to exploit their behavior for better processes. Clues are ignored. Should we? Profitability through Simplicity, June 20, 2023
39. Malhotra, Girish: Considerations to have an excellent environmentally friendly and economic chemical process? Profitability through Simplicity, August 28, 2023
40. Malhotra, Girish: Marriage of Science and Technology in Active Pharmaceutical Ingredient (API) Manufacturing and their Formulations: Is it for real? Profitability through Simplicity, September 21, 2023
41. Malhotra, Girish: Process Simplification and Net Zero: Capitalizing on Physical and Chemical Properties of Reactants and Intermediate, Profitability through Simplicity, August 20, 2024
42. Malhotra, Girish: The Good, The Bad, and The Ugly (1) Complexities of the Pharmaceutical Manufacturing, Profitability through Simplicity, April 9, 2018, Contract Pharma June 2018 Vol. 20 No.5
43. Review of Continuous Process for Modafinil, Continuous Processing in the Chemical and Pharmaceutical Industry II, 2009 AIChE Annual Meeting, November 10, 2009,Nashville, TN.
44. Malhotra, Girish: Analysis of API (Omeprazole): My perspective, Poster Session: Pharmaceutical Engineering, 2009 AIChE Annual Meeting, November 11, 2009, Nashville, TN.
45. Malhotra, Girish: Alphabet Shuffle: Moving From QbA to QbD - An Example of Continuous Processing, Pharmaceutical Processing, February 2009 pg. 12-13
46. Malhotra, Girish: EXPLOITATION OF QBD ELEMENTS FOR A BATCH/CONTINUOUS PROCESS https://www.slideserve.com/elia/exploitation-of-qbd-elements-for-a-batch-continuous-process International Pharmaceutical Academy October 19-20, 2009
47. Malhotra, Girish: Chemicals tell us how to exploit their behavior for better processes. Clues are ignored. Should we? Profitability through Simplicity, June 20, 2023
48. Malhotra, Girish: Use of Tradition, Creativity, Imagination and Technology Innovation in Propofol Manufacturing, Profitability through Simplicity, March 21, 2024
49. Malhotra, Girish: USP 11,267,798 B2: Manufacture of Piperine (1) An Excellent Teaching Tool, Profitability through Simplicity, June, 17, 2024