Fine/specialty chemicals and its family members such as additives, flavors and fragrances and active pharmaceutical ingredients have a commonality. They use similar/same chemicals and equipment for their synthesis for their production but are separated by end use and application. Their differentiation starts from how the same and/or different raw materials are reacted to produce the desired intermediates and products. They also have different quality standards and expectations.
Solvent/water reduction per kilo of the product has always been a part of the process design but not emphasized. Discussion here centers around the opportunities chemists and chemical engineers have to reduce the solvent/water use and simplify processes. My discussion is based on actual experience of how by capitalizing and augmenting the reactivity and method of addition of chemicals, reactions can be optimized and can result in significant reduction of reaction facilitators (solvent/water). Discussion is not influenced by any regulatory, non-profit and for profit organization.
Among the organic chemicals which includes petrochemicals, fine/specialty chemicals, active pharmaceutical ingredients (API) and their formulations have the highest emissions per kilo of product (1). In recent years “Net Zero” (2) has become a mainstream topic. There is conversation but an effort on how we can reduce the solvent use for the production of APIs and their formulations, a subset of fine/specialty chemical product classification that cure diseases is least discussed. With emphasis on lowering emissions per kilo, process developers, when it comes to development have to act and react differently from what we are taught or practice. Solvent recovery and reuse is not enough or sufficient to get to “Net Zero”. Solvent use/reduction is critical for our planet. Creativity and imagination is needed. Volumes can be written on the subject.
We are taught fundamentals of physical properties of the chemicals used and produced. How these can be used to reduce/minimize solvent use in various chemical synthesis are not taught in our universities and colleges. At least we were not taught. They are learnt and experienced on the job during development, scale-up, commercialization of the developed processes. During process development out of the box thinking is required. They are on the job training and developer’s EUREKA moments. Collective creativity (3, 4, 5, 6) helps to optimize processes and reduces solvent use.
Some of the methods to optimize and reduce solvent use could be called magical tricks but they are not. They are pure and simple exploitation and manipulation of physical and chemical behavior. Physical properties of chemicals tell and teach us of their social behavior (7). It is up to us to decide how we can and are able to exploit them to create excellent processes.
Diazotization is a chemical reaction that every chemist and chemical engineer is taught in Organic Chemistry and is used as an illustration. This reaction is about 100 years old and has been the building block of most Dyes in Germany (8) and worldwide. It is also used to produce many other products. Learnings of this and/or similar reactions can be incorporated and implemented to many other chemical reactions. Other examples are reviewed (3, 4, 5, 6).
In the illustrated reaction an amine is converted to its diazonium salt which is reacted further with appropriate molecule to create the desired intermediate/product. Focus here is on the diazonium salt production (Eq. 1).
Aromatic Amine+ 2HCl + Sodium Nitrite ---> Diazonium Salt + H2O + NaCl (1)
Diazo formation reaction is exothermic. To contain the exotherm, i.e. prevent any explosion or run away reaction, it recommendation has been to conduct it at low (0-5°C or lower) temperatures. In early process development reaction exotherm was generally controlled by adding ice to the reaction. This diluted the reaction mass. This was due to unavailability of jacketed reactors or heat exchangers. Reaction product, generally a dye, was filtered and filtrate disposed in river streams as it was the simplest thing to do (3). Impact of effluents on water, fauna and soil was not a consideration. These came later. Heat exchangers were tremendous help in that effort.
About seventy years ago Maumee Chemicals, Maumee Ohio developed a continuous (9) diazotization process for one of its products. This reaction was carried out at 35-40°C., quite an anomaly from the tradition of those and earlier days. This minimized the water/solvent use and improved the productivity of the process. Due to cost considerations hydrochloric acid was the acid of choice. Company commercialized many other chemistries that were novel for their time and were way ahead of even present day conservation considerations.
Illustration of exploitation of mutual behavior of chemicals, reaction mechanism and kinetics is illustrated using Diazo reaction (Eq. 1). Amines are generally a basic chemical. To convert an amine to a diazo salt, it is reacted with an acid. Resulting product is subsequently is reacted with sodium nitrite to produce the respective diazonium salt. Equations 2 & 3 illustrate the reaction mechanism of the diazo reaction. It is acknowledged in most organic chemistry books (10). This sequence can be simulated in the laboratory and in pilot plant.
RNH2 + HCl ⎯> RNH2.HCl (Eq. 2)
RNH2.HCl + NaNO2 +HCl ⎯> RN2Cl +2H2O + NaCl (Eq. 3)
Amine reacts with hydrochloric acid to produce a hydrochloride (Eq. 2) with subsequent reaction with nitrous acid (generated by sodium nitrite and acid reaction) to produce a diazo compound (Eq. 3) that is reacted with a chemical to produce the desired product. This reaction sequences can be capitalized on in a plant by sequential feeding of raw materials, controlling the exotherm and reaction residence time.
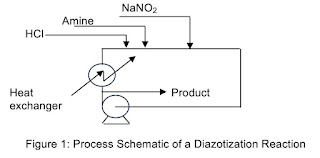
In the reaction step (Eq. 2), the formed hydrochloride is unstable. However, it is converted to produce the diazo product instantaneously as it comes in contact with nitrous acid. Yield of the diazo product is almost 100%. For conservation “Instantaneous reaction” is the key and is manageable. Addition sequence, capitalizing on heat of reaction and equipment scheme are the key for the success. Figure 1 is an illustration of the process.
Theoretically one mole of hydrochloric acid is needed to convert the amine to its hydrochloride and an additional mole of acid is needed to react with sodium nitrite to produce nitrous acid which produces the diazo. As illustrated in Figure 1 by adding slightly excess than two moles of acid, excess of acid assures the hydrochloride formation, assures mixing and formation of nitrous acid to produce the desired diazo compound. Reaction is carried out in a circulating pipe with an inline heat exchanger of proper material of construction. Slight excess of sodium nitrite is needed. They are considerably less than the stoichiometry mentioned in many patents, too many to cite.
Reaction exotherm is controlled by in-line cooling, place and way the chemicals are added to the reaction system and the residence time.
Again, nature of chemicals, how they react and act is the key. Similar addition schemes can be used by the chemists and chemical engineers to create other excellent processes. They can produce active pharmaceutical ingredients, a subset of fine/specialty chemicals and many other organic products. Every chemist and chemical engineer who has mastered their chemistry and process development traits well will totally understand value of such addition methods and processes.
Chemicals share/tell their mutual behavior with us. We have the opportunity to take advantage of them. However, due to tradition we are afraid to step out of the PLAY box to be different.
There are many other situations, where exploiting mutual behavior of chemicals especially as liquids, can be used to simplify organic syntheses. Reaction mass of most syntheses are liquid or a slurry. Liquid/solution are the preferred phase over slurries. Ways and methods to capitalize on social behavior of chemicals have been reviewed (3, 4, 5, 6, 11,12,13, 14, 15, 16,17) and in many other publications. Again, it is up to chemists and chemical engineers to be creative. Many might not believe but such processes based on capitalizing physical and mutual behavior of chemicals used and produced are possible. Unless they are explored, we would not know their value. They are economic and have the highest financial return, a basic premise of great business.
Girish Malhotra, PE
EPCOT International
1. Sheldon R.A. The E factor 25 years on: the rise of green chemistry and sustainability, Green Chemistry https://pubs.rsc.org/en/content/articlelanding/2017/gc/c6gc02157c/unauth#!divAbstract , 2017, 19, 18-43 Accessed February 17, 2021
2. Burke, J. What does net zero mean? https://www.greenbiz.com/article/what-does-net-zero-mean, May 2, 2019 Accessed April 27, 2021
3. Malhotra, Girish: Active Pharmaceutical Ingredient Manufacturing: Nondestructive Creation De Gruyter April 2022 Accessed May 24, 2023
4. Malhotra, Girish: Chemical Process Simplification: Improving Productivity and Sustainability, John Wiley & Sons, February 2011 Accessed May 24, 2022
5. Malhotra, Girish: Chapter 4 “Simplified Process Development and Commercialization” in “ Quality by Design-Putting Theory into Practice” co-published by Parenteral Drug Association and DHI Publishing© February 2011 Accessed May 24, 2022
6. Malhotra, Girish: Research Report: Strategies for Improving Batch or Creating Continuous Active Pharmaceutical Ingredient (API) Manufacturing Processes, March 2017
7. Malhotra, Girish: Sociochemicology May 30, 2013 Accessed January 13, 2023
8. Diazonium Compound https://en.wikipedia.org/wiki/Diazonium_compound
9. Continuous Process https://bit.ly/2Rp3Xlu
10. L. F. Fieser & M. Fieser: Organic Chemistry, Third Edition, Reinhold Publishing Company 1956
11. Malhotra, Girish: Improving APIs & Formulation: Are You Harnessing the Power of Liquids? https://www.linkedin.com/pulse/improving-apis-formulation-you-harnessing-power-liquids-malhotra April 23, 2023 Accessed May 24, 2023
12. Malhotra, Girish: Focus on Physical Properties To Improve Processes: Chemical Engineering, Vol. 119 No. 4 April 2012, pgs. 63-66 Accessed May 24, 2023
13. Malhotra, Girish: Process Simplification and The Art of Exploiting Physical Properties, Profitability through Simplicity, March 10, 2017
14. Malhotra, Girish: Art and Science of Chemical Process Development & Manufacturing Simplification, AIChE May 17, 2023 Accessed May 24, 2023
15. Blog Profitability through Simplicity Accessed May 20, 2023
16. Malhotra, Girish: Review of Continuous Process for Modafinil, Continuous Processing in the Chemical and Pharmaceutical Industry II, 2009 AIChE Annual Meeting, November 10, 2009, Accessed May 20, 2023
17. Malhotra, Girish: Analysis of API (Omeprazole): My perspective, Poster Session: Pharmaceutical Engineering, 2009 AIChE Annual Meeting, November 11, 2009 Accessed May 20, 2023